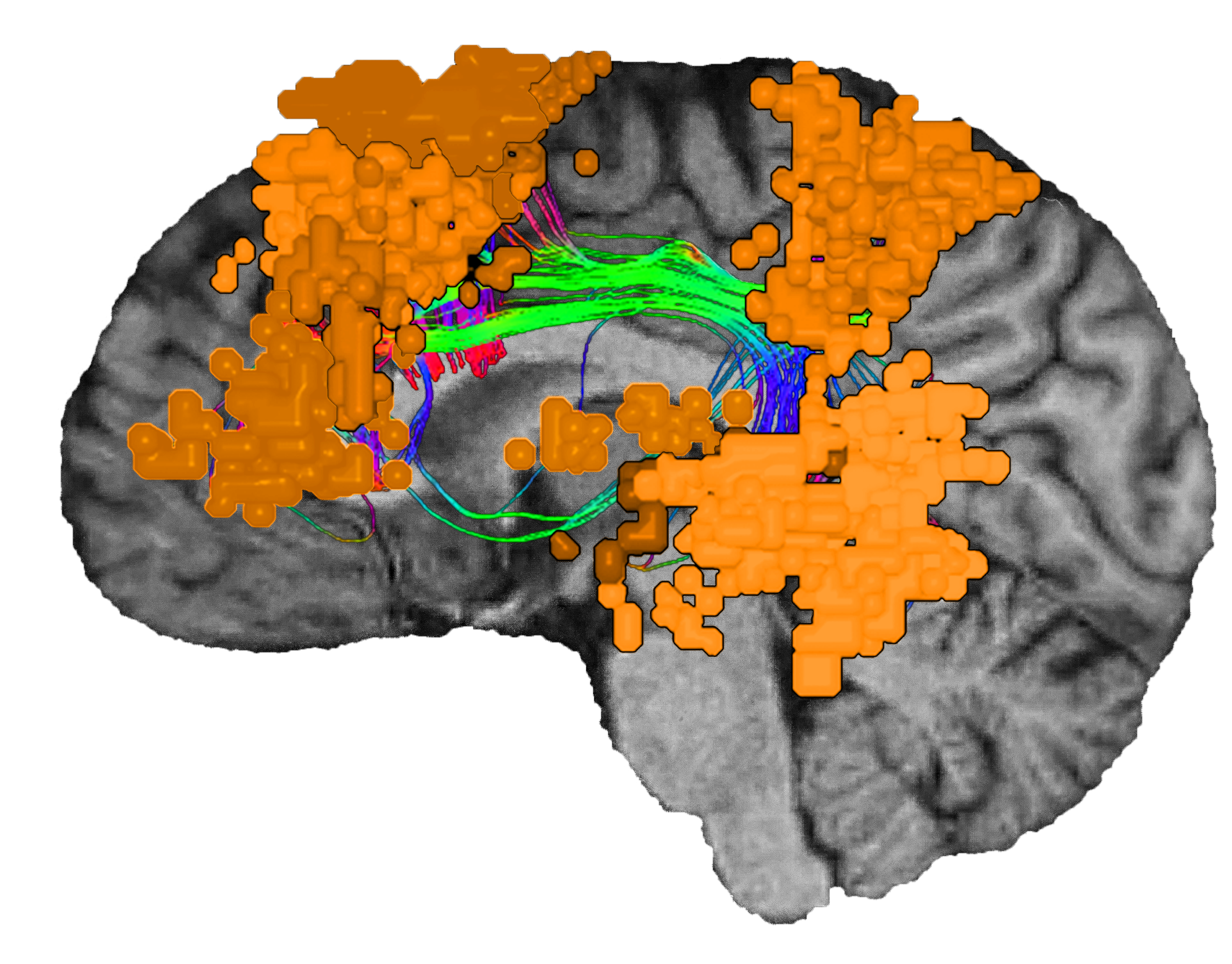
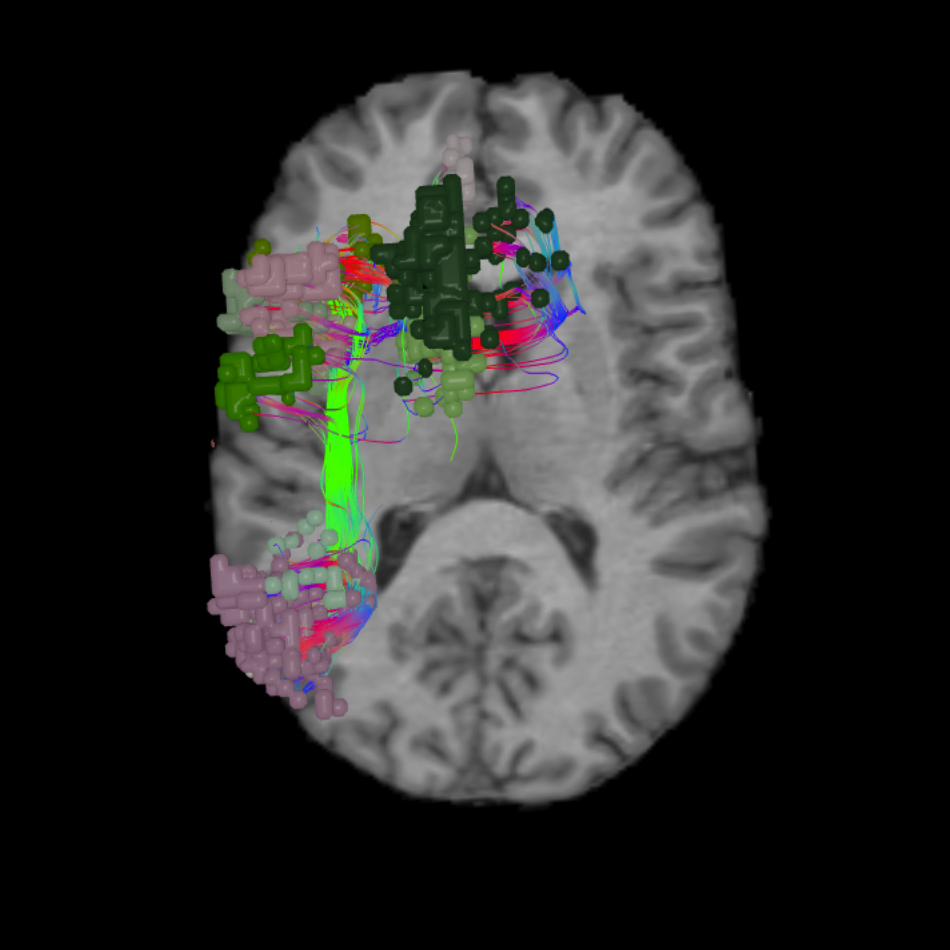
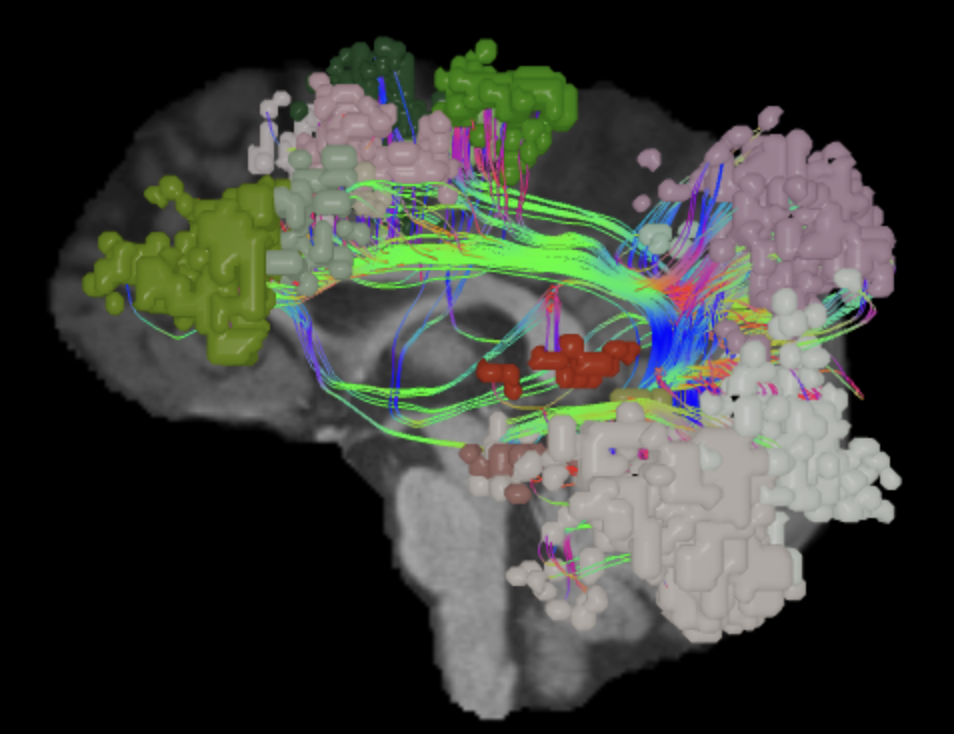
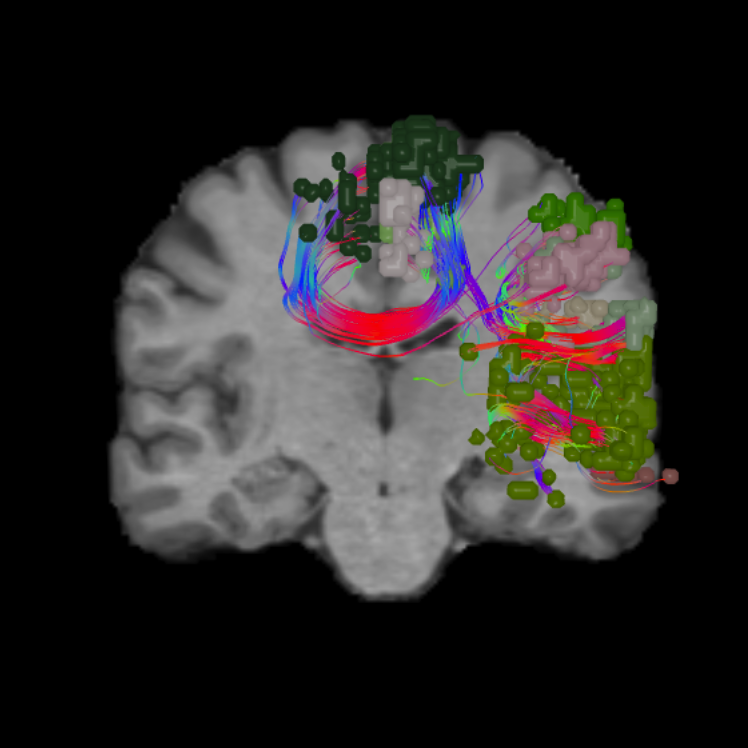
Damage to functional areas and white matter tracts13 of the language system can result in problems with language, speech, word association, recognition and familiarity,22 or naming.23 Injury to this system has also been implicated in neurological conditions and psychiatric disorders.
Stroke
In stroke patients, the location of the stroke plays a large factor on the severity of the damage. Since the language system is widespread in the left hemisphere, strokes in the lower left parts of the brain may cause greater amounts of language or speech dysfunction. Functional areas TE1a, TE1m, and TE1p are located in this region, and due to their implication in language processing tasks,24 can result in difficulty with language following a stroke.
Language disorders following a stroke often include alexia, agraphia, and aphasia.25 Alexia is a type of sensory aphasia where an individual loses the ability to read,25 whereas agraphia is the loss of writing skills.25 Difficulty writing may manifest from problems with language processing, visual perception, motor planning,25 or a combination of factors.
Aphasia is the loss or impairment of verbal communication,25 but it has been demonstrated to cause problems with writing and reading, as well as listening or speaking. Aphasia is most commonly caused by stroke, but may also result from a lesion in the left hemisphere25 or damage to the white matter tracts connecting regions of language in the brain — including the articulate fasciculus (AF), inferior longitudinal fasciculus (ILF), and the inferior fronto-occipital fasciculus (IFOF).6
However, in some cases, aphasia may also be a progressive, degenerative disease. Chawluk and colleagues26 have also suggested that progressive aphasia may occur without dementia and as a separate clinical syndrome from Alzheimer’s disease.26
Semantic dementia
Semantic dementia (SD) is the result of frontotemporal lobar degeneration27 typically occurring asymmetrically in the left hemisphere.28 Ventrorostal temporal lesions are also associated with the degeneration of white matter tracts connecting language areas29 that may lead to SD. Patients with semantic dementia have shown lower functional connectivity in the left uncinate fasciculus (UF), which has been previously linked to language functionality.30,31
SD is characterized by progressive aphasia — particularly anomia — and difficulty comprehending the meaning of words.27 Progressive stages of the disease have also shown atrophy near the contralateral temporal pole and medial prefrontal cortices which can result in social and behavioral changes.28
Unfortunately, early identification of SD can be difficult with standard clinical and neuropsychological evaluations.27 Using neuroimaging and task-related fMRI studies may offer opportunities to identify biomarkers of disease progression28 that may lead to earlier diagnoses and possible therapies.27
Epilepsy
Epilepsy has been shown to impact semantic memory in patients with varying outcomes based on whether epilepsy occurs in the left or right temporal lobe.32 Functional areas TGd and STGa play an important role in semantic memory,33,34,35 which will require further research on their possible implications in epilepsy.
Drane and colleagues36 found that patients experiencing right temporal lobe epilepsy (TLE) and post-surgical deficits demonstrate difficulty with face recognition and familiarity judgements.36 Since functional areas of the language system are primarily located in the left hemisphere, patients with left TLE unsurprisingly demonstrate difficulty with face naming.36 However, they did not exhibit problems with recognition or familiarity.36
Performing surgical treatment for TLE may reduce seizures, but it relies on accurate lateralization of the TLE. Resting-state fMRI has been useful for mapping connectivity and determining left or right lateralized TLE. Morgan and colleagues identified an area in the ventral lateral nucleus of the right thalamus with functional connectivity to the hippocampi that may be an indicator of lateralization and distinguish left from right TLE subjects.37 Predicting hemispheric language dominance helps surgeons preserve the greatest amount of language function following an operation.
Connectomics offers the opportunity to define key functional language areas and their white matter connections with great specificity. In addition to understanding where primary areas of language reside in each patient’s brain, continued research may also lead to more specific models of semantic memory and the localization of seizures to improve post-surgical outcomes.32
Since functional areas and white matter tracts of the language system make up a highly interconnected system, developing patient-specific connectomic atlases will aid in diagnosing patients13 and providing greater opportunity for therapy or treatment.