What is the ventral attention network?
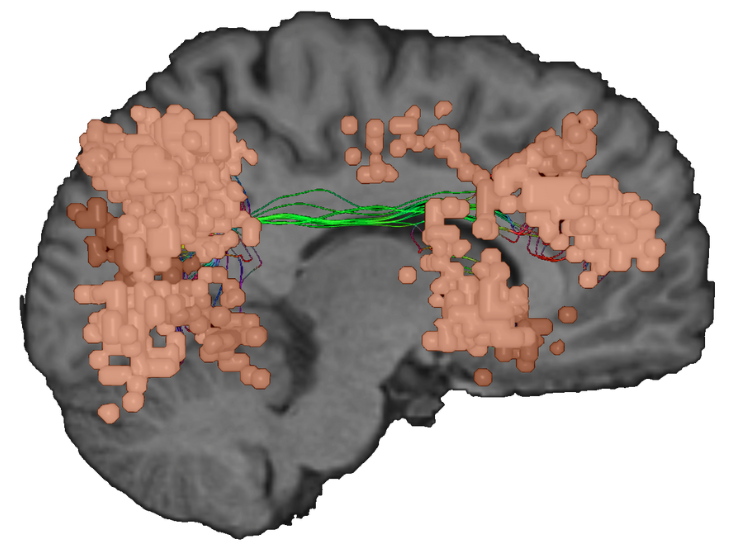
The VAN is a unilateral, typically right hemispheric brain network responsible for swapping our focus between various tasks and responding to unexpected cues, making it pivotal to daily function1.
As the brain’s radar, the ventral attention network, (sometimes referred to as the visuospatial attention network) is frequently activated in relation to the other major attentional network, the Dorsal Attention Network (DAN). Where the DAN could be considered “the lens”, responsible for focussing and guiding attention toward a salient stimulus, the VAN plays a more pronounced role in pivoting our attention when new information is introduced.
Imagine walking through a garden on a hot summer’s day. While walking through this garden your attention may focus solely on smelling the roses, however far more likely is that your attention would be grabbed along the way by new, more salient information: the humming of bees, the heat on the back of your neck, and other cues which arise on your journey that need to be interpreted to fully understand your situation. The VAN is the brain network enabling this interpretation, and by communicating with other brain networks such as the limbic system, visual, and auditory networks, enables our taking in of our complete surroundings.
The human brain receives sensory input constantly, and it is the VAN’s role to juggle our attention between this information and ensure that we do not neglect to respond to important spatial, sensory, or cognitive information. Indeed, the inability to facilitate this is broadly classified as “Spatial Neglect”.
To return to the analogy, as you continue to walk through the garden, your friend appears and passes you a ball. Though your limbic and auditory networks are still sending cues about the smell of the flowers and sounds of the bees, your attention must now shift and respond to the unexpected cue of the incoming ball. The VAN acts to respond to this new stimulus, recruiting the sensorimotor network and switching your attention towards catching the ball. After this conflict is resolved, the VAN switches your attention back to your previous state of reverie, enabling a smooth transition of high and low attention-demanding states.